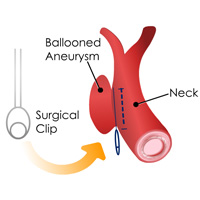
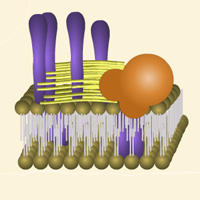
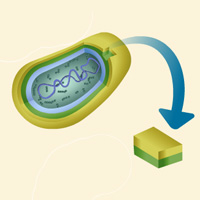
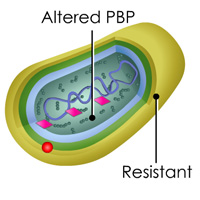
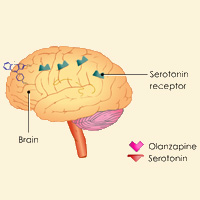
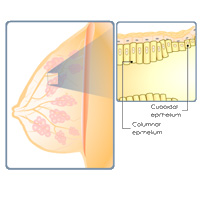
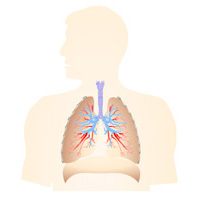
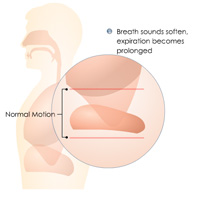
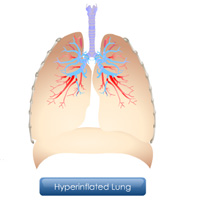
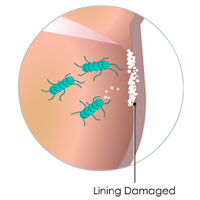
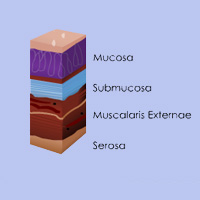
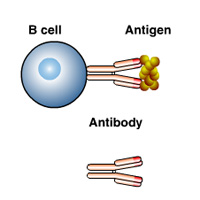
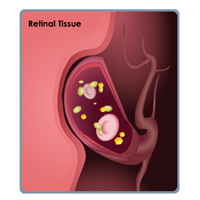
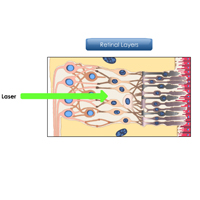
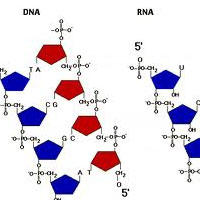
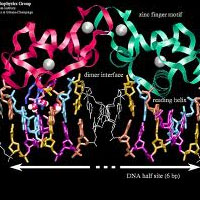
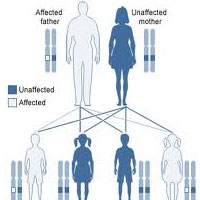
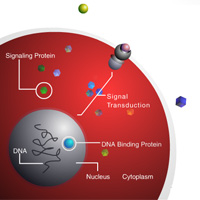
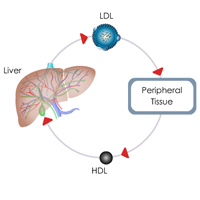
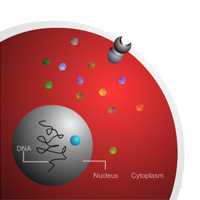
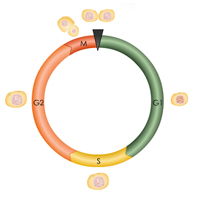
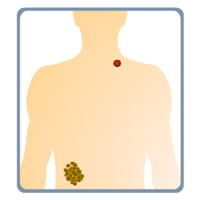
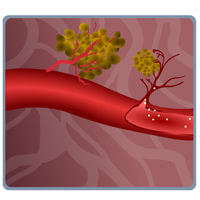
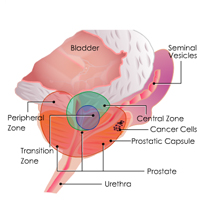
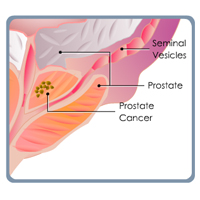
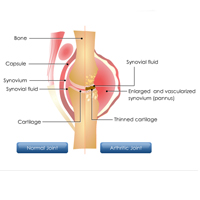
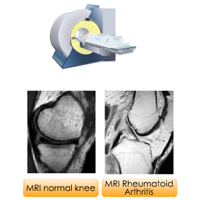
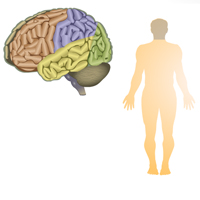
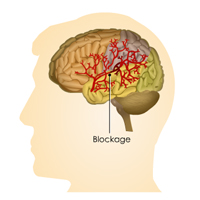
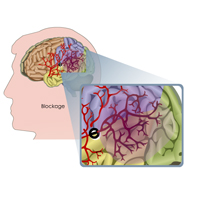
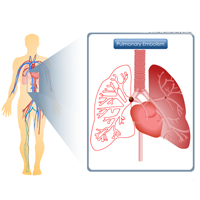
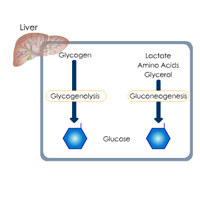
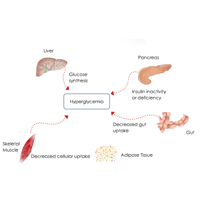
Complete List of Archived Courses and Professional Training Programs
Have Questions? Contact Us:
Use the form below to request more information about our programs. You can also request a quote for group training or an invoice to generate a company purchase order. For quotes or invoices, please include the course title(s) and the number of students.


































![Certification Training Clinical Trials: 21 CFR Part 50 Subpart B - Informed Consent of Human Subjects (HSP) [Part 1 of 2]](../images/cimages/ELM-909.png)
![Certification Training Clinical Trials: 21 CFR Part 50 Subpart B - Informed Consent of Human Subjects (HSP) [Part 2 of 2]](../images/cimages/ELM-910.png)